Medulloblastomas are the most common primary malignant brain tumors of childhood. They are responsible for about 10% of all brain tumors in children. Rarely, they also occur in adults, but here they account for less than 1% of all brain tumors. The most common localization is in the cerebellum. The standard therapy consists of surgery and, depending on age, subsequent chemotherapy and/or radiotherapy, which are individually adapted. The prognosis and individual therapy is also highly dependent on the tumor subtype.
What is a medulloblastoma?
Medulloblastoma is an embryonal tumor, meaning that it arises from immature, undifferentiated cells of the central nervous system (CNS) and grows very rapidly. Medulloblastomas are highly malignant. Meanwhile, genetic analysis of the tumors has identified 4 molecular subgroupes of medulloblastoma, which differ in their molecular genetic features and also have different prognoses.
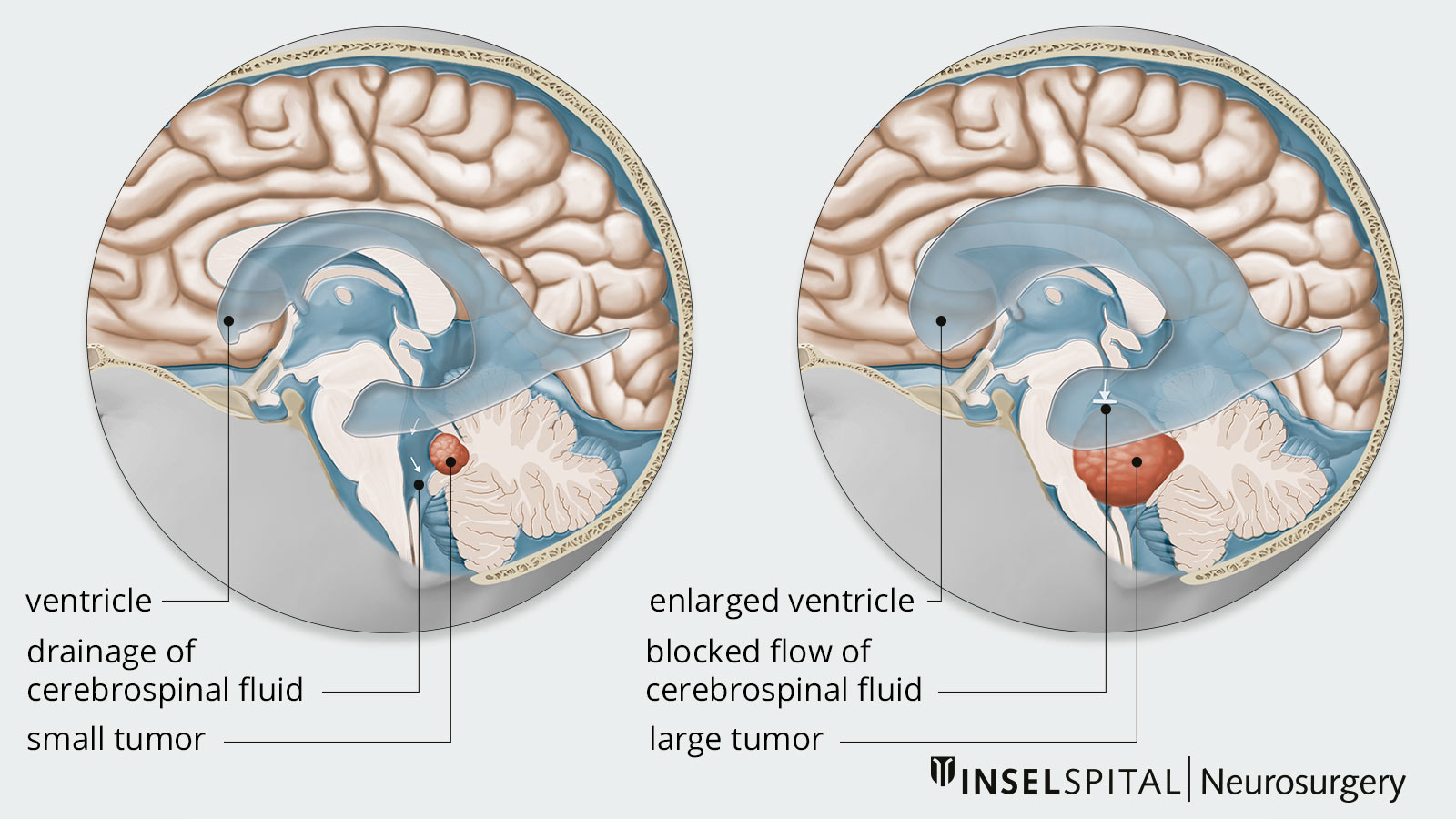
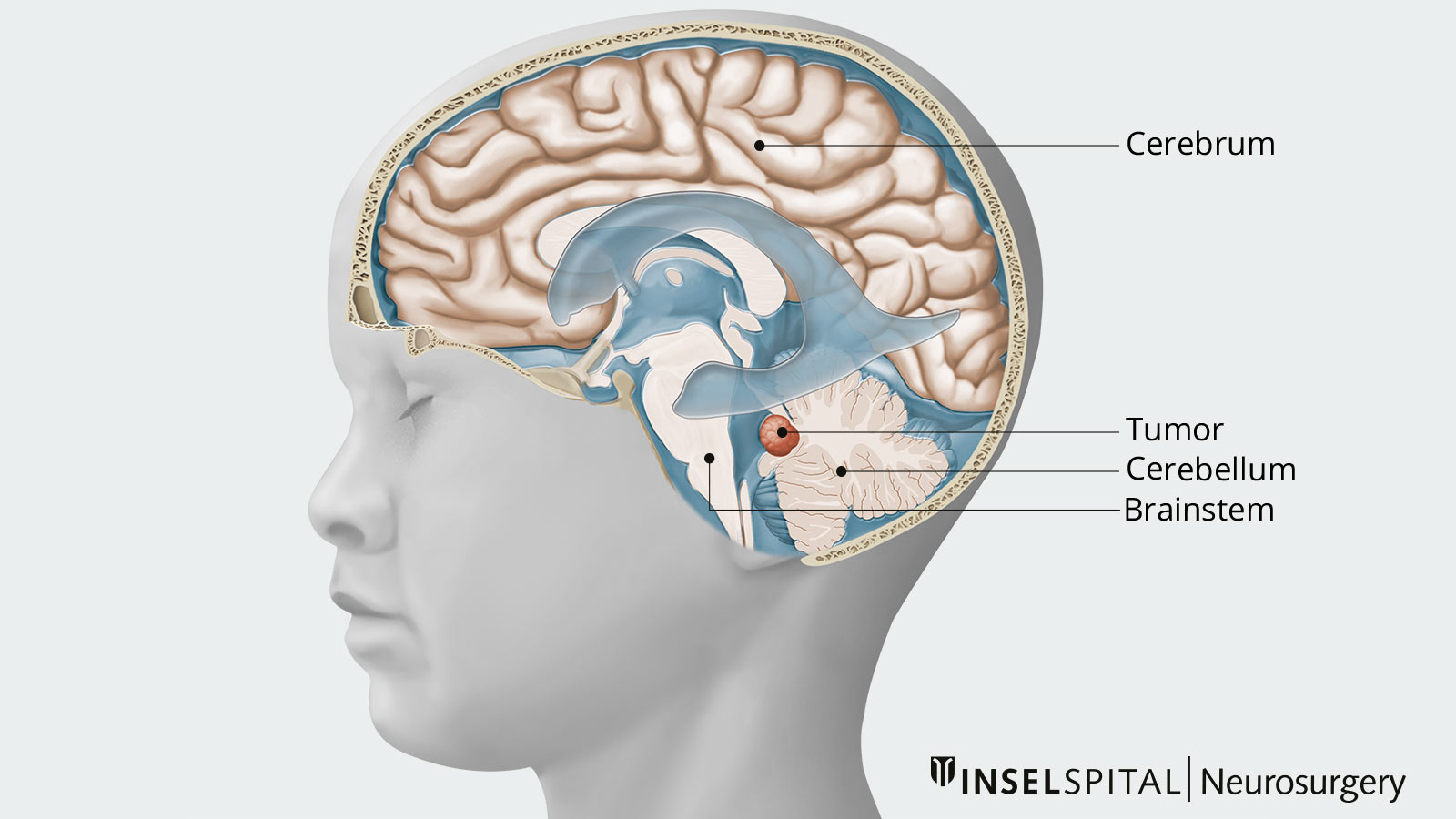
Medulloblastomas are typically found in the posterior fossa in the area of the fourth ventricle (fluid-filled cavity). Spread of tumor cells via the cerebrospinal fluid (CSF) to other brain regions or the spinal cord is possible. Most commonly, medulloblastoma locally infiltrates the cerebellum and brainstem. Spread to the cerebrum is more commonly found in adults.
Due to the rapid and aggressive growth of medulloblastomas, adjacent vital structures can be displaced and damaged. Furthermore, depending on its exact location, the tumor can cause a blockage of the CSF flow.
What is the cause of a medulloblastoma?
Medulloblastoma is primarily caused by a spontaneous, unpredictable degeneration of cells of the nervous system. The cause of this sudden and uncontrolled cell growth is still unclear. However, genetic factors play a recognizable role. Less than 5% of cases are associated with hereditary diseases (so-called cancer predispositions). These include:
- Gorlin-Goltz syndrome
- Rubinstein-Taybi syndrome
- Turcot syndrome
- Li-Fraumeni syndrome
Who is affected by a medulloblastoma?
The age of onset for medulloblastomas varies with the subtype of the tumor. They typically occur in two age groups: in infants between 3–4 years and in older children between 8–10 years. In adults, the tumors are very rare. 10–50% of medulloblastomas have already spread at the time of diagnosis. The most common are so-called drip metastases. These spread along the subarachnoid space or the spinal axis. Metastases outside the nervous system are much rarer. These occur in around 5% of patients.
What symptoms does a medulloblastoma cause?
Usually, symptoms develop within a short time because of the rapid growth. When the cerebellum and the fourth cerebral ventricle are affected by the medulloblastoma, dizziness with nausea and vomiting, coordination problems and movement disorders such as gait unsteadiness occur.
However, it can also happen that the brain stem is affected, which can lead to cranial nerve failures. This manifests itself in visual disturbances, double images, facial paralysis or similar.
When cerebrospinal fluid flow is obstructed, hydrocephalus results from the accumulation of CSF with a corresponding increase in intracranial pressure. Patients then suffer from symptoms such as headaches, nausea, vomiting or pronounced fatigue and drowsiness. In addition, confusion and personality changes may also occur. In young children, an above-average and fast-growing head is sometimes noticeable. This macrocephaly is a sign of increasing cerebrospinal fluid and increasing intracranial pressure. Affected children appear very sleepy or react irritably.
Spinal metastases can lead to pain and sensorimotor deficits due to the space-occupying effect on the spinal cord.
How is a medulloblastoma diagnosed?
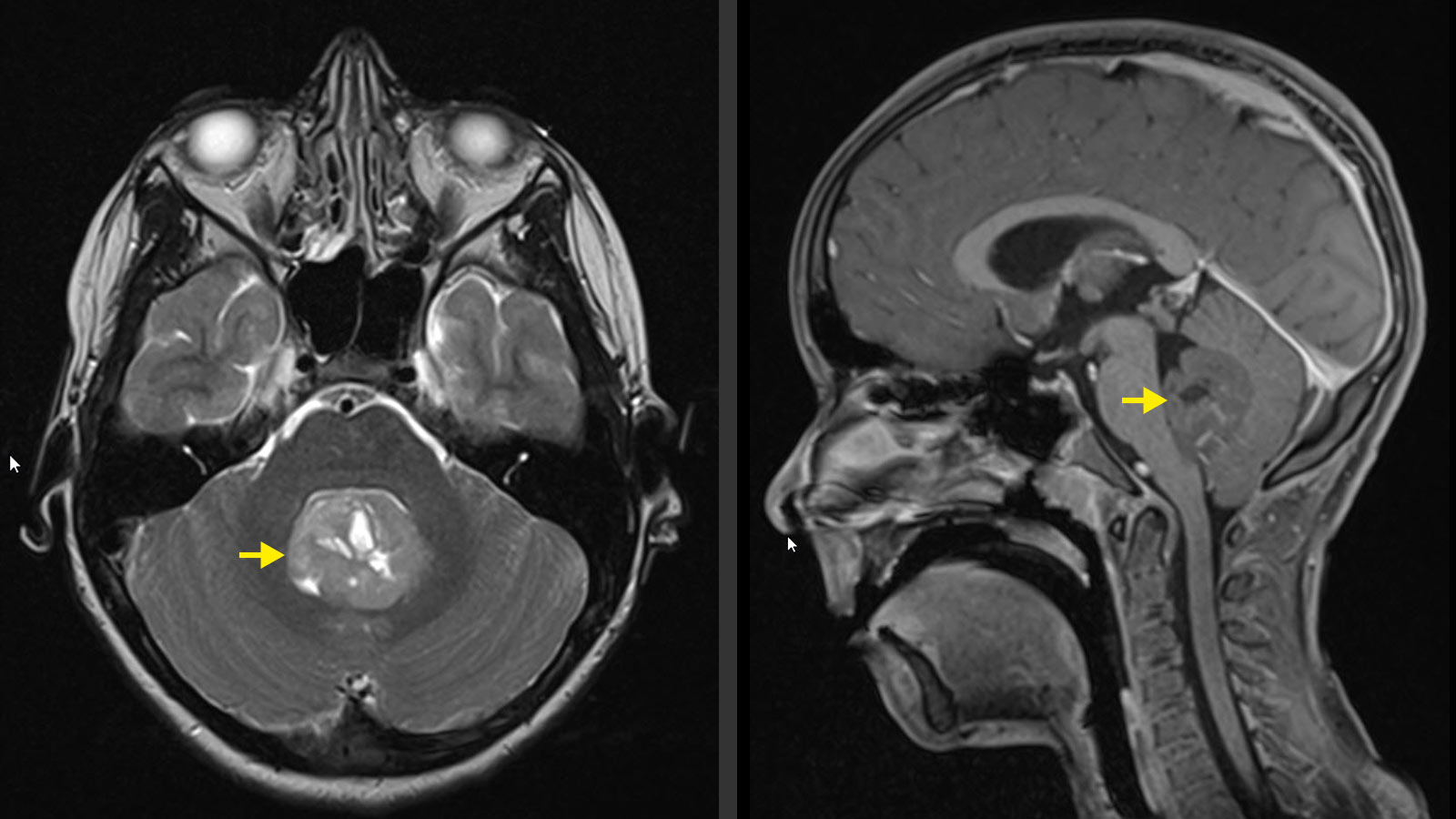
On computed tomography (CT), medulloblastoma typically looks bright and takes contrast. Often, calcium deposits are seen in the tumor. However, magnetic resonance imaging (MRI) is considered the imaging gold standard, and should include the spine in addition to the skull to rule out possible metastases throughout the central nervous system.
In young children, if the fontanelles are still open, indications can already be seen in the ultrasound sonography of the head.
In addition to imaging, a lumbar puncture is performed to examine the cerebrospinal fluid, as tumor cells can occasionally already be detected here.
However, an exact diagnosis can only be made after histological and molecular genetic evaluation of the surgically removed tumor tissue. Advanced techniques such as DNA methylome profiling and next-generation sequencing (NGS) have significantly improved the diagnosis and classification of medulloblastomas by making it possible to characterize the tumours at the molecular level *, *.
Classification
According to the new WHO classification, medulloblastomas are divided into 4 molecular subgroups * based on genetic and molecular characteristics. This distinction has implications for prognosis and treatment *:
- WNT-activated: these tumors are associated with a very good prognosis and show few genetic alterations. They are characterized by the presence of mutations in the beta-catenin gene and monosomy 6.
- SHH-activated: This group includes tumours characterized by activation of the Sonic Hedgehog pathway. SHH tumors associated with TP53 mutations have a poorer prognosis.
- Group 3 and Group 4: These groups are less well characterized than WNT and SHH, but show different biological and clinical behaviors, which influences treatment and prognosis.
In addition, further subtypes must be differentiated: four subtypes of SHH and eight subtypes of group 3 and group 4.
Therapy options
The therapy of choice is microsurgical resection of the tumor as completely as possible. The extent of resection is decisive here and complete removal of the tumor tissue is associated with a better prognosis. The planning of the operation and subsequent treatment is carried out in close cooperation with an interdisciplinary team consisting of neurosurgeons, neuroradiologists, neuropathologists, oncologists and radiation oncologists.
Post-operative therapy by means of craniospinal radiation and/or chemotherapy depends on individual criteria such as the subtype of the tumor, the patient's age, the patient's general state of health, the metastasis and, last but not least, the patient's tolerance to the therapeutic measures. For children under 3 years of age, intensified chemotherapy is often used to delay the need for radiotherapy, as this is highly toxic and can have long-term side effects.
Collaboration between pediatric neurosurgeons, radiation oncologists and pediatric oncologists is critical to ensure optimal treatment.
The new discoveries of subgroups and subtypes may lead to changes in treatment in the future, such as individualized therapy with different treatment protocols. Especially in children, this may lead to the minimization of long-term therapy-associated side effects. *,*, *
Prognosis
The chances of success vary greatly depending on the subgroup and subtype of the tumor. In children under 3 years of age, radiotherapy has severe side effects (neuropsychological deficits, endocrinopathies), so that only postoperative chemotherapy is used in such young patients. The primary goal of surgical treatment is safe minimally invasive yet maximally radical removal of the tumor with restoration of unrestricted CSF outflow. If the patient's own CSF outflow cannot be restored, a ventriculoperitoneal shunt may be required. In this case, the CSF is drained via a catheter under the skin into the abdominal cavity.
Why you should seek treatment at Inselspital
Depending on the location of the medulloblastoma, there is a certain risk of damaging neighboring brain structures during surgery. To keep this risk of complications as low as possible, we use the latest technical procedures such as neuronavigation and intraoperative neuromonitoring. These allow our neurosurgeons to work with maximum precision and safety and still achieve the maximum tumor resection.
-
Juraschka K, Taylor MD. Medulloblastoma in the age of molecular subgroups: a review. J Neurosurg Pediatr. 2019 Oct 1;24(4):353-363. doi: 10.3171/2019.5.PEDS18381.
-
Taylor MD, Northcott PA, Korshunov A, Remke M, Cho YJ, Clifford SC, Eberhart CG, Parsons DW, Rutkowski S, Gajjar A, Ellison DW, Lichter P, Gilbertson RJ, Pomeroy SL, Kool M, Pfister SM. Molecular subgroups of medulloblastoma: the current consensus. Acta Neuropathol. 2012 Apr;123(4):465-72. doi: 10.1007/s00401-011-0922-z.
-
Louis DN, Perry A, Wesseling P, Brat DJ, Cree IA, Figarella-Branger D, Hawkins C, Ng HK, Pfister SM, Reifenberger G, Soffietti R, von Deimling A, Ellison DW. The 2021 WHO Classification of Tumors of the Central Nervous System: a summary. Neuro Oncol. 2021 Aug 2;23(8):1231-1251. doi: 10.1093/neuonc/noab106.
-
Ramaswamy V, Remke M, Bouffet E, Bailey S, Clifford SC, Doz F, Kool M, Dufour C, Vassal G, Milde T, Witt O, von Hoff K, Pietsch T, Northcott PA, Gajjar A, Robinson GW, Padovani L, André N, Massimino M, Pizer B, Packer R, Rutkowski S, Pfister SM, Taylor MD, Pomeroy SL. Risk stratification of childhood medulloblastoma in the molecular era: the current consensus. Acta Neuropathol. 2016 Jun;131(6):821-31. doi: 10.1007/s00401-016-1569-6.
-
Maier H, Dalianis T, Kostopoulou ON. New Approaches in Targeted Therapy for Medulloblastoma in Children. Anticancer Res. 2021 Apr;41(4):1715-1726. doi: 10.21873/anticanres.14936.
-
Lutz K, Jünger ST, Messing-Jünger M. Essential Management of Pediatric Brain Tumors. Children (Basel). 2022 Apr 2;9(4):498. doi: 10.3390/children9040498.
Further reading
- Robinson GW, Rudneva VA, Buchhalter I et al. Risk-adapted therapy for young children with medulloblastoma (SJYC07): therapeutic and molecular outcomes from a multicentre, phase 2 trial. Lancet Oncol. 2018;19:768-784.
- Harbaugh R, Shaffrey CI, Couldwell WT. Neurosurgery Knowledge Update. Thieme; 2015:984.
- Greenberg MS. Handbook of Neurosurgery. Thieme; 2016:1664.
- The Neurosurgical Atlas: Medulloblastoma (free sign up).