Cavernomas are relatively common vascular malformations of the brain and occur in about 1 in 200 to 1 in 600 people. Many cause no symptoms and are discovered incidentally during an MRI scan; in these cases, the prognosis is usually reassuring. The most common symptom is epileptic seizures, while the overall risk of bleeding is low, though it may be higher for cavernomas located in the brainstem. The decision regarding whether treatment is necessary is made on a case-by-case basis and is evaluated at Inselspital during interdisciplinary team meetings.
What are cavernomas?
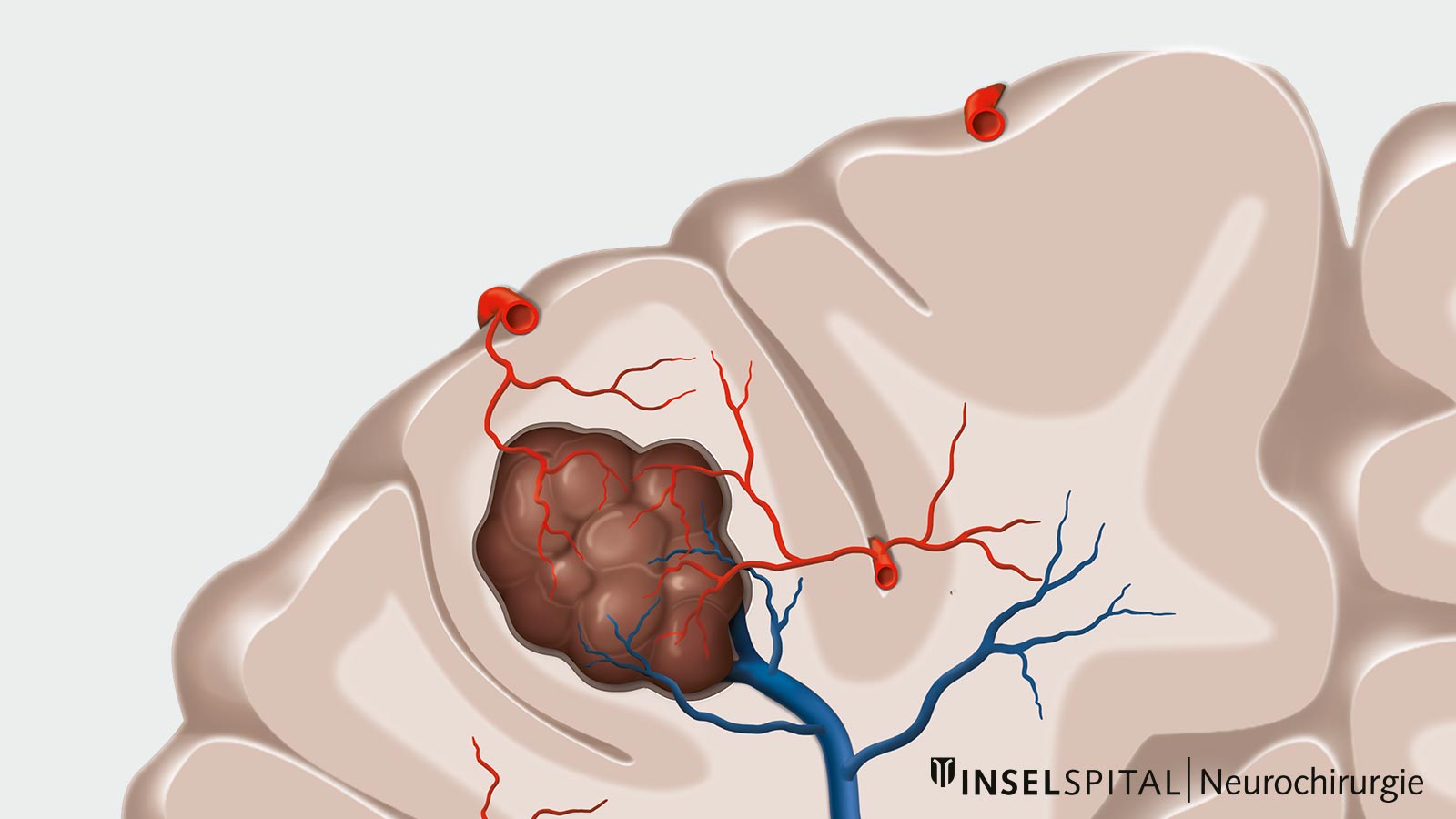
Cavernomas (also known as cerebral cavernous malformations, cavernous angiomas, or cavernous hemangiomas) are vascular malformations in the brain. They consist of abnormally dilated, thin-walled capillary spaces that often cluster together in a grape- or mulberry-like pattern.
Unlike arteriovenous malformations, blood flow in cavernomas is slow, which is why they are generally not visible in conventional angiography. Their size can vary from a few millimeters to several centimeters.
The walls of these vessels are thin and fragile. As a result, small amounts of blood can leak into the surrounding brain tissue over time. This blood and its breakdown products can irritate the brain tissue and trigger symptoms such as epileptic seizures or headaches.
However, many cavernomas cause no symptoms and are discovered incidentally during an MRI scan.
Key Features
- Not visible in vascular imaging studies such as catheter angiography (so-called “angiographically occult”)
- 70–80% of cavernomas are located in the cerebral hemispheres
- 15–20% are located in the brainstem
- About 5% are located in the spinal cord
- About 40% cause no symptoms and are discovered incidentally
How common are cavernomas?
Cavernomas are among the most common vascular malformations of the brain.
- The prevalence is approximately 0.16% based on incidental MRI findings and up to 0.5% in autopsy studies, corresponding to about 1 in 200 to 1 in 600 people.
- The detection rate is approximately 0.56 per 100,000 inhabitants per year and is likely increasing due to the growing use of magnetic resonance imaging (MRI).
- Cavernomas are therefore discovered incidentally more often today than in the past. Most of these incidental findings never cause symptoms.
What symptoms can a cavernoma cause?
Approximately 20–40% of cavernomas are incidental findings during magnetic resonance imaging (MRI). These cavernomas do not cause any symptoms and therefore have not been noticed.
The most common symptom of a cavernoma is epilepsy. Other symptoms can vary widely and usually depend on the location and size of the cavernous vascular malformation.
Smaller hemorrhages or thromboses often occur within the cavernoma. Therefore, additional symptoms may include paralysis, sensory disturbances, speech or visual disturbances, dizziness or headaches.
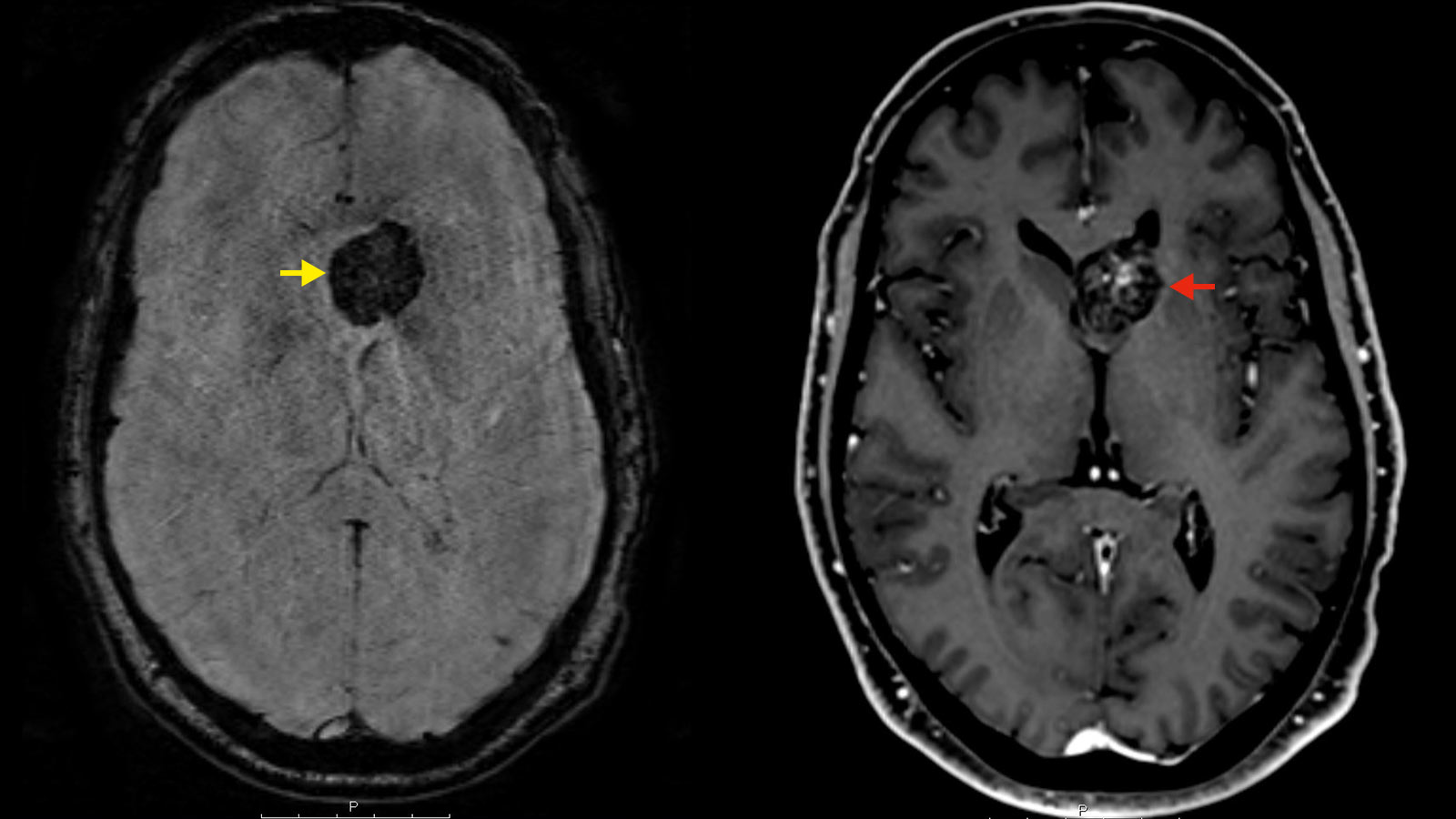
How is a cavernoma diagnosed?
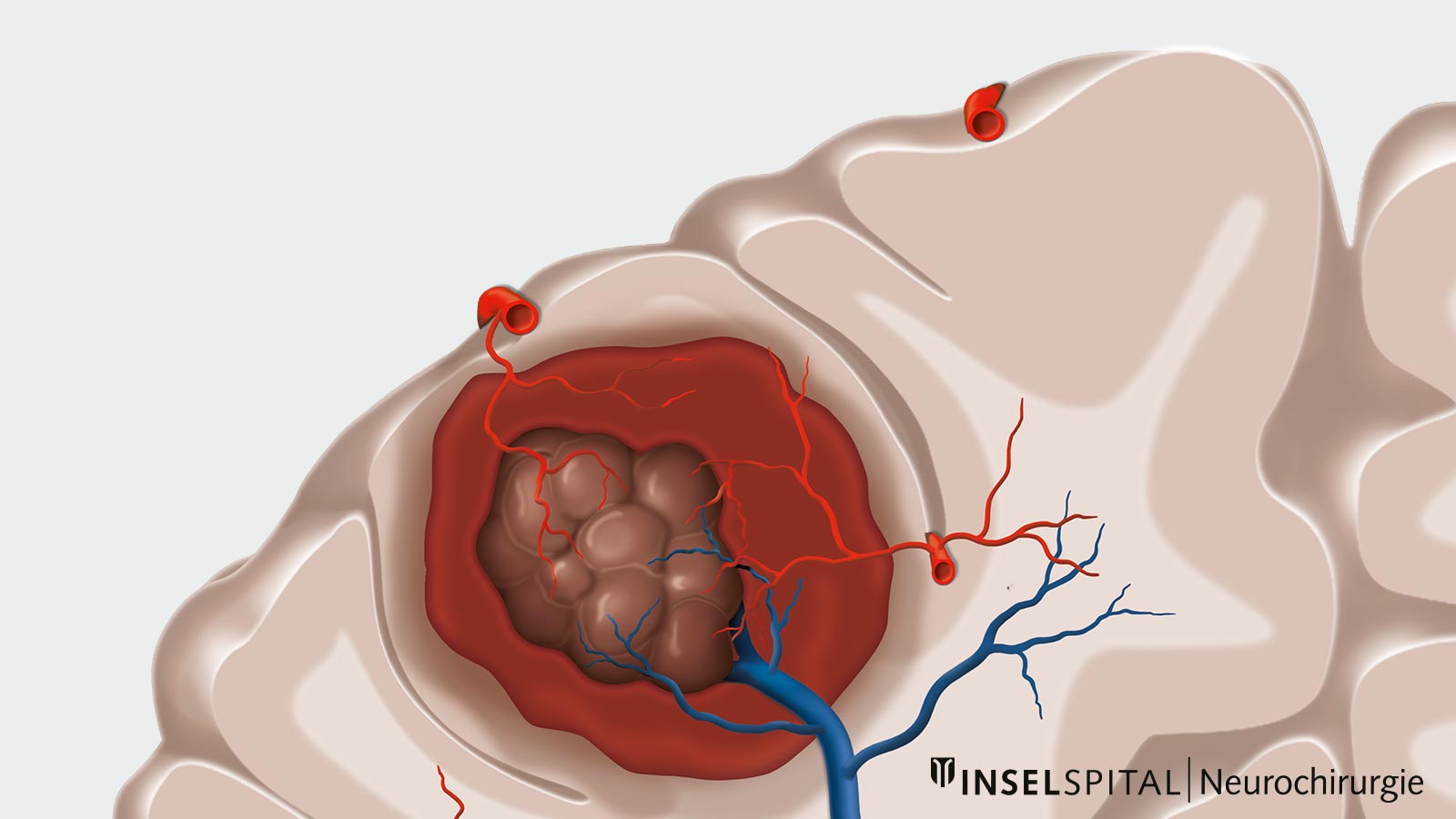
Typical of cavernomas is their dynamic behavior. Although cavernomas do not have large arteries or veins inside and blood flow is slow and low, they can still cause bleeding. Usually, cavernomas do not cause large and dramatic hemorrhages,but minor hemorrhages to nearby areas often occur when a cavernoma empties or ruptures. Therefore, cavernomas are almost always surrounded by scarring, the so-called gliosis, and blood degradation products that can be visualized radiologically. * *
The diagnosis of a cavernoma is made during an MRI examination with the administration of contrast medium. With the help of special sequences (SWI sequences), blood degradation products can be visualized and a statement made as to whether or not a cavernoma has already bled once or not.
Does a cavernoma need surgery?
Whether or not cavernomas should be surgically removed depends on numerous factors and must be decided for each patient on a case-by-case basis. Important factors here are symptomatology and localization. * Compared to cavernomas in the brainstem, cavernomas in the cerebrum are much more harmless in their course. ** Furthermore, cavernomas are relatively often (in about 60% of cases) associated with venous anomalies, which may also influence the surgical procedure.
Surgery is usually only recommended for cavernomas that have become symptomatic. The closer a cavernoma is to functionally important areas, the earlier it becomes symptomatic. This is the reason why cavernomas are a special case in surgical neurosurgery: Surgery must often be performed in the immediate proximity of important functional areas. Therefore, a very careful indication for surgery is needed.
Why you should seek treatment at Inselspital
At Inselspital we have established one of the largest cavernoma programs in Switzerland. Since setting up our specialized cavernoma database, we have systematically recorded and monitored over 400 patients. This experience makes our center a leading center of excellence for the treatment of cavernomas.
Clinical Care
- We offer the full spectrum of treatment—from observation to microsurgical resection with state-of-the-art intraoperative monitoring.
- Our surgical approach combines neuronavigation, intraoperative neurophysiological monitoring, microsurgical techniques, and—when appropriate—awake surgery.
- We have particular expertise in the surgery of brainstem cavernomas, supported by specialized monitoring protocols.
Research and International Collaboration
- Our cavernoma database enables us to systematically investigate the natural course and risk factors of this condition.
- A study from our center reported an average annual bleeding risk of approximately 2.3%. *
- Our research also showed no significant association between the risk of bleeding and the use of antiplatelet agents, an important contribution to the international discussion on the safety of antithrombotic therapies.
- Our data were also incorporated into an international collaboration published in The Lancet Neurology, which suggests that antithrombotic therapy may reduce the risk of cavernoma hemorrhage. *
- We actively monitor the development of new pharmacological and interventional therapeutic approaches.
-
Clatterbuck RE, Eberhart CG, Crain BJ, Rigamonti D. Ultrastructural and immunocytochemical evidence that an incompetent blood-brain barrier is related to the pathophysiology of cavernous malformations. J Neurol Neurosurg Psychiatry. 2001;71:188-192.
-
Horne MA, Flemming KD, Su IC et al. Clinical course of untreated cerebral cavernous malformations: a meta-analysis of individual patient data. Lancet Neurol. 2016;15:166-173.
-
Vernooij MW, Ikram MA, Tanghe HL et al. Incidental findings on brain MRI in the general population. N Engl J Med. 2007;357:1821-1828.
-
Bervini D, Jaeggi C, Mordasini P, Schucht P, Raabe A. Antithrombotic medication and bleeding risk in patients with cerebral cavernous malformations: a cohort study. J Neurosurg. 2018 Jun 8;130(6):1922-1930.
-
Zuurbier SM, Hickman CR, Tolias CS et al. Long-term antithrombotic therapy and risk of intracranial haemorrhage from cerebral cavernous malformations: a population-based cohort study, systematic review, and meta-analysis. Lancet Neurol. 2019;18:935-941.
-
Al-Shahi Salman R, Berg MJ, Morrison L, Awad IA, Angioma ASAB. Hemorrhage from cavernous malformations of the brain: definition and reporting standards. Angioma Alliance Scientific Advisory Board. Stroke. 2008;39:3222-3230.
-
Zimmerman RS, Spetzler RF, Lee KS, Zabramski JM, Hargraves RW. Cavernous malformations of the brain stem. J Neurosurg. 1991;75:32-39.
-
Bozinov O, Hatano T, Sarnthein J, Burkhardt JK, Bertalanffy H. Current clinical management of brainstem cavernomas. Swiss Med Wkly. 2010;140:w13120.