Meningiomas are slow-growing tumors that originate in the meninges. At around 35%, they are among the most common tumors of the central nervous system (CNS) and occur more frequently after the age of 50. More than 90% of meningiomas are benign. When meningiomas grow continuously and reach a size that compresses the brain and causes symptoms, they should be treated.
What is a meningioma?
The brain and spinal cord are surrounded by three protective membranes called meninges. Tumors that arise from the cobweb skin (arachnoid membrane) of the meninges are called meningiomas. Meningiomas are the most common benign intracranial tumors.
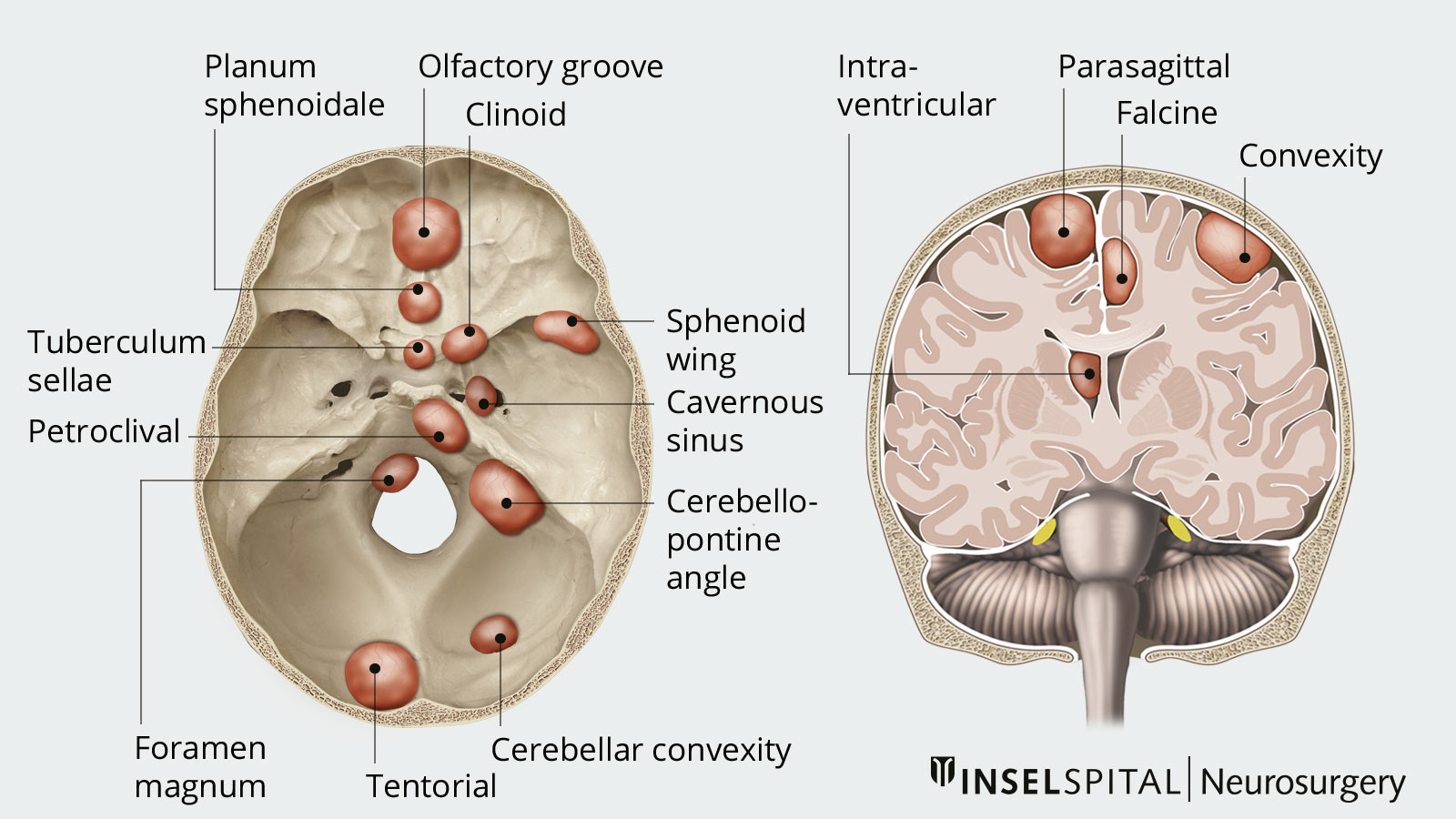
Depending on its location, a meningioma causes different symptoms.
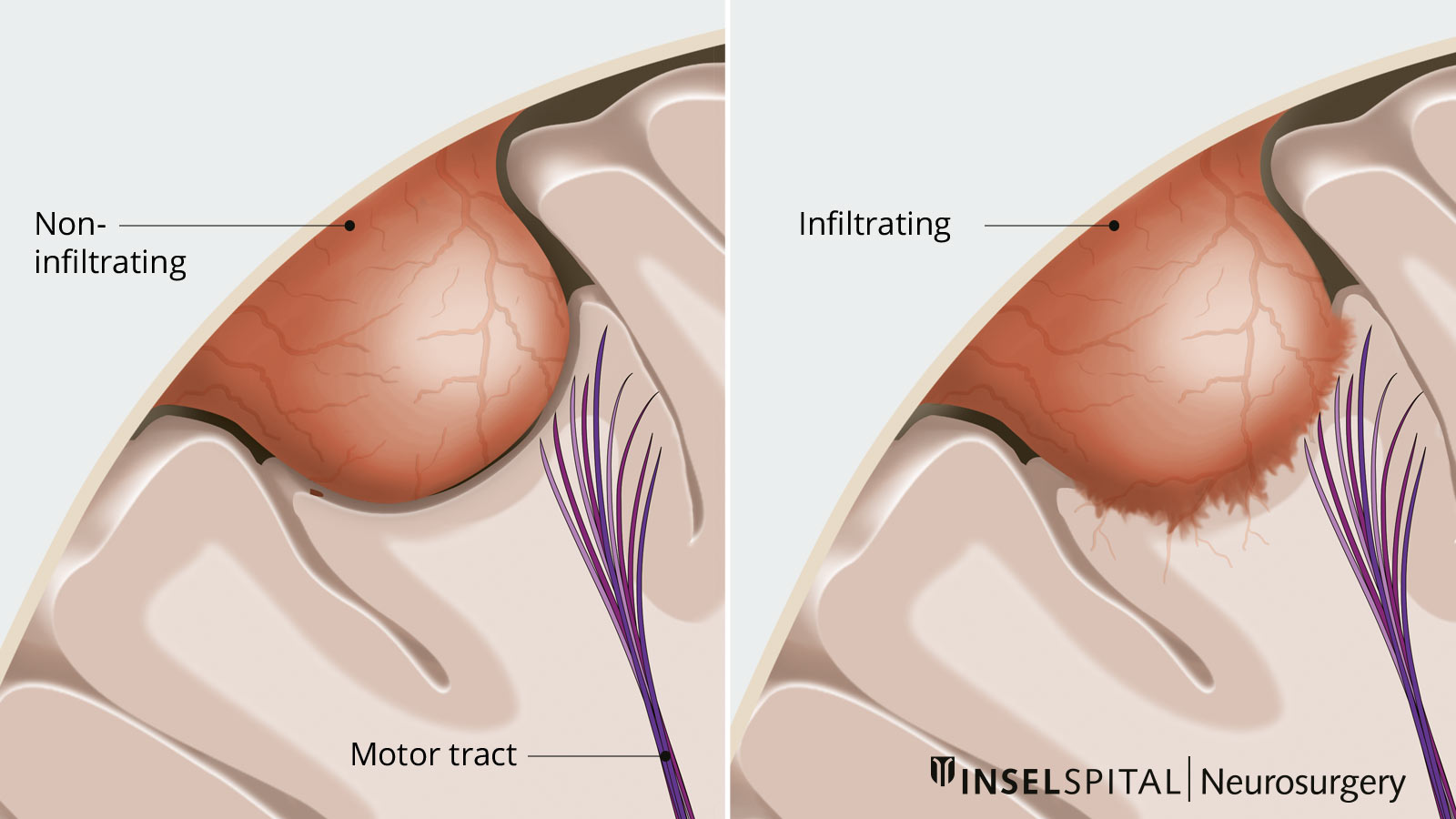
Meningiomas usually grow very slowly, but in rare cases can infiltrate surrounding tissue and metastasize. Although meningiomas are usually benign, they sometimes go undetected for a long period of time due to their insidious, displacing growth, which can lead to severe damage and disability.
What are the causes of meningiomas?
Ionizing radiation is currently considered the most significant known risk factor for the development of meningiomas. People who have been exposed to high doses of radiation have a significantly increased risk of developing a meningioma over the course of their lives. This includes, for example, survivors of the atomic bombings, who have been reported to have a 6- to 10-fold increased risk, as well as patients who have undergone radiation therapy to the head and neck.
People with the inherited condition neurofibromatosis type 2 also have an increased risk of developing meningiomas.
Frequency and risk factors
- most frequent tumors of the CNS
- biggest risk factor: ionizing radiation
- increased occurrence from the age of 50 onwards
How are meningiomas classified?
According to the World Health Organization (WHO), meningiomas are classified into three grades based on histopathological criteria:
- WHO Grade I: approx. 70–80%
- WHO Grade II: approx. 15–25%
- WHO Grade III: approx. 1–3%
WHO Grade I meningiomas are the most common type and are generally benign. They usually grow slowly and have a relatively low recurrence rate after complete removal.
WHO Grade II meningiomas often grow more rapidly, can invade surrounding tissue, and carry an increased risk of recurrence.
WHO Grade III meningiomas are rare and malignant (anaplastic). They grow aggressively, recur more frequently, and usually require a combination of surgery and radiation therapy.
What symptoms do meningiomas cause?
Meningiomas usually grow slowly. As they grow, they can compress the surrounding brain tissue or nearby nerves and blood vessels, thereby causing symptoms. The symptoms that occur depend primarily on the tumor’s location, size, and rate of growth.
Since meningiomas usually grow slowly, symptoms often develop gradually and increase over time. It is not uncommon for meningiomas to cause no symptoms for a long time and to be discovered incidentally during imaging (incidental finding).
Common symptoms:
- headaches
- epileptic seizures
- vision, smell, or speech disorders
- paralysis or sensory disturbances
What happens if a meningioma is discovered by chance?
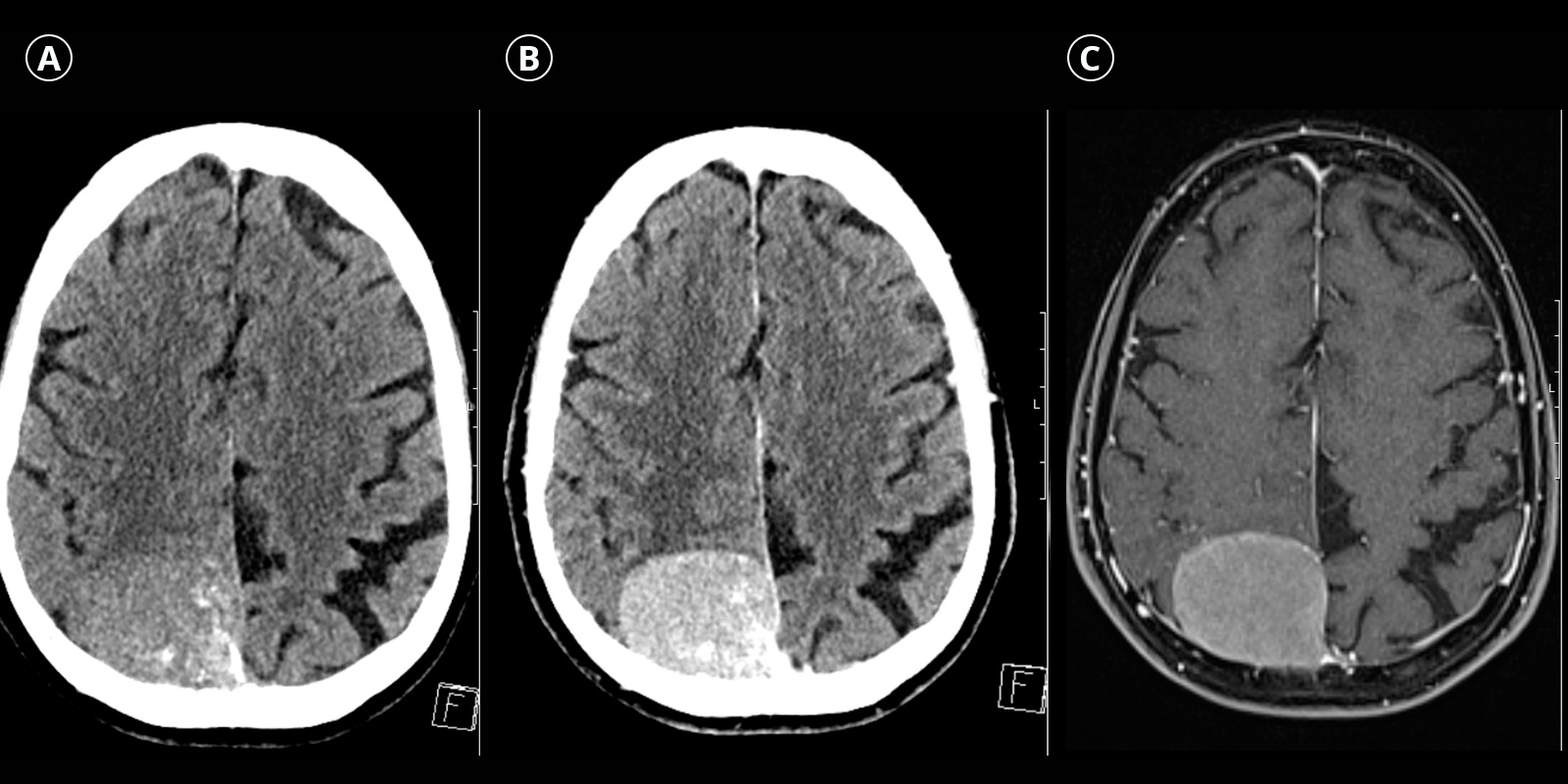
Many meningiomas are discovered incidentally, for example during imaging for nonspecific headaches or following an accident. Such findings are referred to as incidental findings. The diagnosis is often made using magnetic resonance imaging (MRI) or computed tomography (CT).
If the meningioma is small, causes no symptoms, and is not located near functionally important areas of the brain, regular follow-up is often recommended initially. This is usually done through repeated MRI scans, often at yearly intervals at first.
Certain characteristics may indicate an increased potential for growth. These include, among others, a tumor size of more than 2.5 cm, accompanying cerebral edema, or specific signal changes on T2-weighted MRI. In such cases – or if the tumor is located in an unfavorable position – even an incidentally discovered meningioma may require treatment.
How are meningiomas diagnosed?
Imaging Techniques
Meningiomas are usually diagnosed during the evaluation of neurological symptoms or as an incidental finding during imaging performed for other reasons.
In both computed tomography (CT) and magnetic resonance imaging (MRI), meningiomas typically appear as well-defined, extra-axial tumors—that is, tumors that primarily develop outside the brain tissue. A characteristic feature is extensive contact with the dura mater, accompanied by displacement or compression of the adjacent brain tissue.
After administration of contrast medium, meningiomas generally show homogeneous contrast uptake. On CT, calcifications and secondary bony changes may also be detected. On MRI, an outwardly extending dural thickening, known as the dural tail sign, is frequently observed.
How are meningiomas treated?
Small meningiomas that do not increase in size can be treated in a conservative manner. If meningiomas grow continuously or have reached a size that compresses the brain, causes cerebral edema, or becomes symptomatic, they should be treated. Standard therapy is surgery, less common is radiosurgical therapy.
Because of the variable location and size, the decision about the best approach must always be discussed on an individual basis. The optimal treatment therefore depends on the size, location and growth rate of the tumor, as well as on histopathological tissue analysis and the patient's general condition.
Conservative treatment
Patients with asymptomatic incidental findings, small meningiomas or older age are initially treated conservatively. This means that clinical controls including imaging are performed regularly to monitor the progression of the disease. Initial follow-up is performed within 3‒6 months after the first diagnosis. If the findings are stable, the subsequent time intervals between follow-up examinations can be progressively extended.
In various studies, stable meningioma size without growth tendency was observed in about 50‒70% of patients during imaging follow-up over an average period of 2‒5 years. If the meningiomas increase in size or new symptoms develop, a new decision on further therapy should be made.
Operation
The primary therapy for a symptomatic, enlarging meningioma is complete microsurgical removal. If this is possible, it means cure for the patient in most cases. Therefore, the top priority of treatment is to remove the tumor as gently as possible while preserving its function and maintaining a high cosmetic standard.
In this procedure, the meningioma is first reduced in size from the inside so that the border to the neighboring tissue is relieved. Then the tumor is carefully dissected away from the surrounding normal brain tissue under the microscope. The tumor-bearing meninges are usually removed and replaced as well. The interlocking between the normal brain and the meningioma varies in intensity from patient to patient. Some of the tumors have a smooth border, others have infiltrated the brain and are supplied with blood by normal brain vessels.
In these cases, if an important brain area is affected, the operation must be performed under functional monitoring. For this purpose, the latest technical procedure such as neuronavigation with augmented reality and intraoperative neuromonitoring, are used. They enable the neurosurgeon to work with maximum precision and safety under the operating microscope and to make the patient transparent, i.e. to look into tissue.
Preoperative embolization
Meningiomas are often highly vascularized. To minimize bleeding during surgery, it is important to occlude the blood vessels supplying the tumor as early as possible.
In some cases—for example, with certain meningiomas at the base of the skull—these supplying blood vessels are difficult to access during surgery or can only be reached at a later stage. In such cases, a procedure known as endovascular embolization can be performed prior to the actual surgery. During this procedure, the blood vessels supplying the tumor are selectively occluded as part of a minimally invasive vascular procedure. This reduces blood flow to the tumor, which can make the surgery safer in selected cases and minimize bleeding.
Irradiation
In patients with meningioma that is inoperable or only partially operable, radiation therapy is a treatment option with good tumor size control. Radiation therapy is also used as an adjuvant for higher-grade meningiomas or incomplete resection. However, the meningiomas must not have exceeded a certain size for this.
Radionuclide therapy
In difficult cases with gradual disease progression despite surgery and radiation, radionuclide therapy offers another therapeutic option. This involves targeting the tumors with radioactive drugs. The radiopharmaceutical, the radioactive substance, binds to special receptors on the surface of the meningioma, the somatostatin receptors, and achieves a local radiation effect there, thus destroying the tumor cells.
Risks of the procedure
The risk of complications during surgical removal of a meningioma—and, if necessary, during additional radiation therapy—depends on various factors. These include, above all, the size, location, and accessibility of the tumor, as well as the patient’s age and overall health.
For meningiomas in uncomplicated locations, the risk of serious complications is generally low, at less than 2%. For tumors in complex locations or larger tumors, the risk may be higher.
The risks in each individual case depend heavily on the specific situation. We would be happy to discuss these with you personally based on your magnetic resonance imaging (MRI) and explain your personal risk profile.
What happens after the surgery?
The prognosis for a meningioma depends on several factors. The most important factors are the tumor’s location and size, as well as its histological characteristics, i.e., the WHO grade (I–III). Another important factor is whether the tumor could be completely or only partially removed during surgery. The results of the histological examination and—in some cases—molecular biomarkers also play a role in planning further treatment. Based on these findings, the next steps are determined on an individual basis.
WHO grade I and total resection
One control after surgery, depending on the risk, another control after 2.5 and 10 years
WHO grade I and incomplete resection
Annual follow-up and/or radiosurgery
WHO grade II and total resection
Follow-up controls with MRI every 6‒24 months, follow-up irradiation if necessary
WHO grade II and incomplete resection
Annual follow-up and/or radiosurgery
WHO grade III
Follow-up irradation, radionuclide therapy or experimental chemotherapy if necessary, and follow-up controls with MRI every 3‒6 months
Why you should seek treatment at Inselspital
At Inselspital, a tailored treatment strategy is developed for each patient. This takes place at the certified Brain Tumor Center, where an interdisciplinary team collaboratively evaluates the various treatment options and recommends the best possible therapy.
The discussion takes place during a weekly tumor board meeting. There, specialists from the fields of neurosurgery, neurology, neuro-oncology, nuclear medicine, radiation oncology, and pathology consult together on each individual case.
This ensures that all important professional perspectives are incorporated into the treatment decision.