About 2% of all people have a balloon-like enlargement of a cerebral artery – a so-called aneurysm. These aneurysms usually do not cause any specific symptoms and are often discovered as incidental findings. However, each aneurysm can rupture and cause a severe cerebral hemorrhage. Unruptured aneurysms should therefore be checked regularly during MRI examinations. If they remain small and stable, they do not need treatment in most cases. If they grow or show signs of an increased risk of rupture, they should be closed in a microsurgical operation or via a vascular catheter. Here at Inselspital we offer the entire spectrum of aneurysm therapy by renowned experts.
What is an aneurysm and how does it develop?
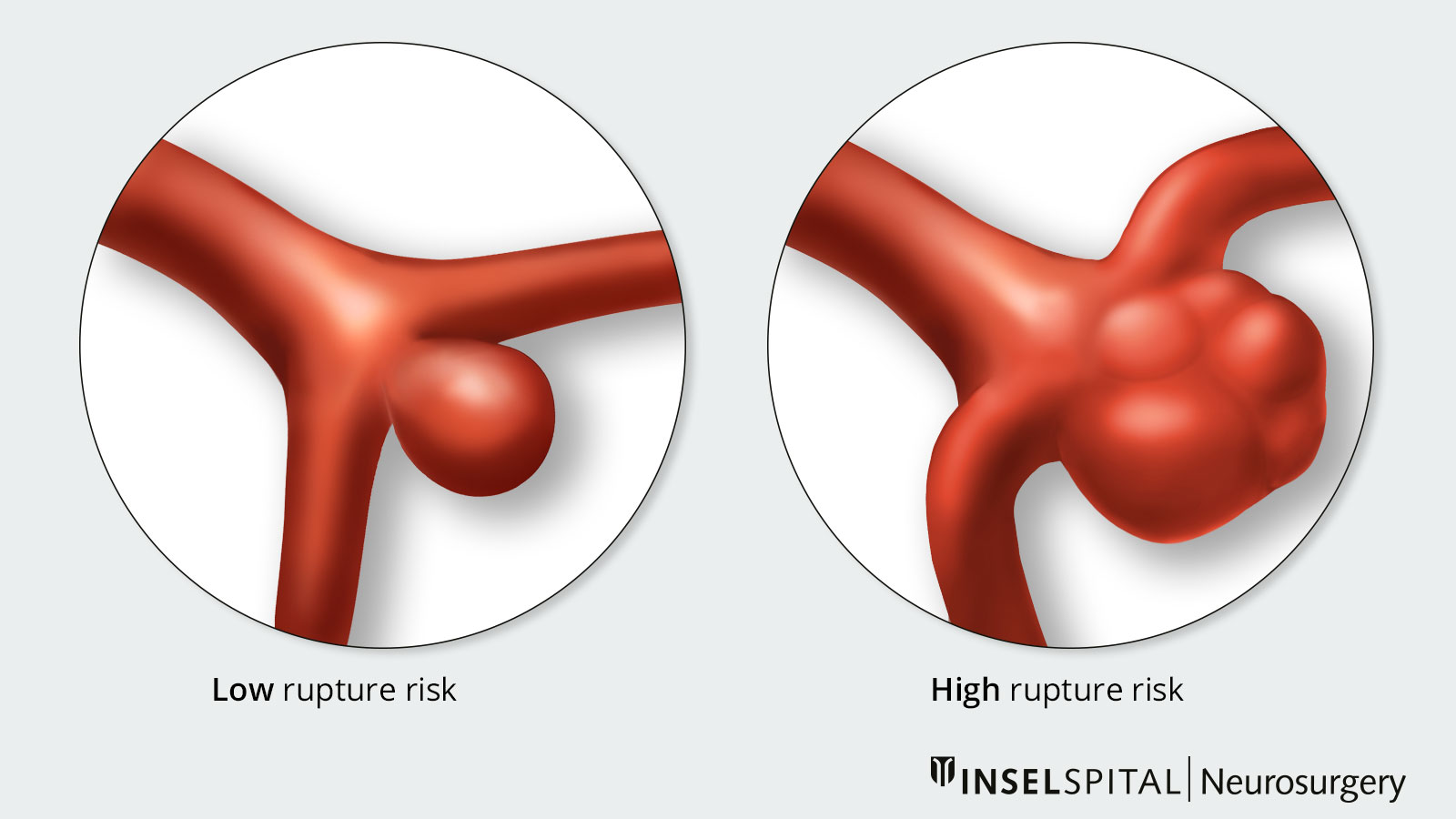
An aneurysm is a balloon-like enlargement of an artery in the brain. It occurs when the vessel wall is weaker in one area and bulges outward.
There can be various reasons for this:
- Congenital weaknesses in the vessel wall
- Risk factors such as high blood pressure, smoking, or family history
- Changes in the vessels over the course of a lifetime, for example due to wear and tear
Most aneurysms cause no symptoms and therefore go unnoticed. They are often discovered by chance during an examination.
An aneurysm becomes dangerous when it ruptures. This can cause sudden and severe bleeding in the brain—a life-threatening emergency.
How common are aneurysms?
Approximately 2% of the population has an aneurysm in the brain, which is often discovered by chance. This figure has been confirmed in several large-scale analyses of angiograms—for example, in a study that evaluated brain MRI images of healthy adults *.
Sometimes higher frequencies of up to 6% are reported. However, these are mostly based on autopsies, in which the advanced age of the individuals examined and the detection of even the smallest vascular irregularities skew the figures upwards. For the general population, therefore, around two in every hundred people have an aneurysm, usually without knowing it.
Incidental finding of an aneurysm – what happens next?
If an aneurysm is discovered incidentally, in most cases we recommend a consultation with one of our specialist neurosurgeons or neuroradiologists. At the neurosurgery clinic at Inselspital, we offer detailed consultations during the aneurysm consultation hours with Prof. Andreas Raabe or Prof. David Bervini.
Interdisciplinary assessment: The findings and medical history are then usually discussed together in the weekly aneurysm board meeting.
Follow-up: In most cases, the board initially recommends regular check-ups.
Treatment in case of risk: If there are clear risk factors for the aneurysm rupturing, we recommend treatment.
The decision is always made on an individual basis. Follow-up checks and possible treatments are carefully weighed up together with the patient. Personal attitude also plays an important role: some people prefer to live with the low risk of rupture, while others prefer the one-time risk of treatment.
Follow-up examinations
In most cases, we recommend an annual follow-up examination using MRI without contrast agent. Even if an aneurysm remains stable for years, there is always a certain risk that it will grow.
However, the frequency of follow-up examinations is adjusted individually, depending on the patient's age and the size and shape of the aneurysm. The optimal calculation of follow-up intervals is a current research topic in neurosurgery and neuroradiology at Inselspital
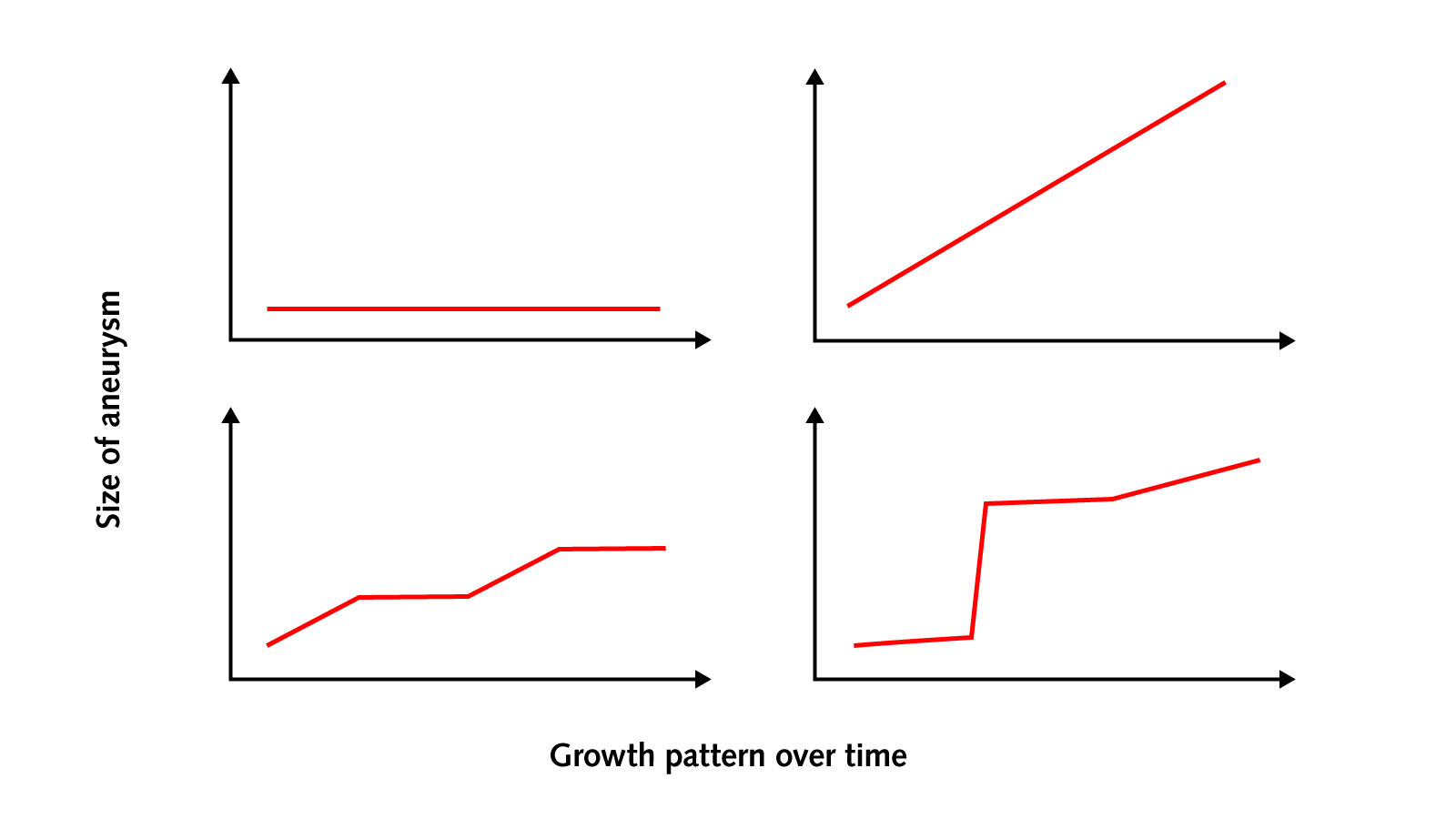
In 2–5 % of patients, the aneurysm grows over time or forms an additional bulge («daughter aneurysm»). Both changes increase the risk of bleeding by about tenfold and are a reason for treatment in most cases.
Further monitoring despite growth is rarely recommended and depends on the individual treatment risk.
Since aneurysms do not have a uniform growth pattern, it is not possible to make a reliable prediction about future risk even after years of stability.
Rupture risk
If an aneurysm in the head ruptures, it is life-threatening:
- Approximately 30% of those affected die from the bleeding or its consequences.
- Less than 20% survive without permanent damage.
This raises an important question: Should an aneurysm be treated preventively before it ruptures? The answer depends on whether the risk of rupture is higher than the risk of treatment.
Studies show:
- Small (<7 mm) aneurysms in the anterior circulation that are discovered by chance have a very low risk of bleeding of about 0.2% per year.
- Other aneurysms, on the other hand, can have a risk of over 10% per year.
It must therefore be carefully considered whether regular monitoring or treatment is more beneficial.
You can find more information on ruptured aneurysms and cerebral hemorrhages on the page Ruptured aneurysm and subarachnoid hemorrhage.
Risk factors
The following factors—individually or in combination—can increase the risk of bleeding and thus influence the decision to treat. If there are no risk factors, the risk of bleeding is less than 0.2 % per year.
Significantly increased risk
- Aneurysm larger than 10 mm
- Growth in size over time (confirmed by MRI)
- Familial clustering: at least two affected first-degree relatives
Increased risk
- Aneurysm larger than 7 mm
- Enrichment of the aneurysm wall with contrast medium during examination
- Familial clustering: at least 2 affected second-degree relatives
- Certain genetic disorders (e.g., polycystic kidney disease)
- Other genetic risk factors
- Previous bleeding from another aneurysm
- Abnormal findings in imaging (e.g., irregular shape, daughter aneurysm, size ratio > 1)
- Location in the posterior circulation
- Multiple aneurysms
- Smoking
- Increased alcohol consumption
- High blood pressure
How is an aneurysm treated?
The goal of treating a brain aneurysm is to completely and permanently isolate the aneurysm from the bloodstream in order to prevent rupture and cerebral hemorrhage.
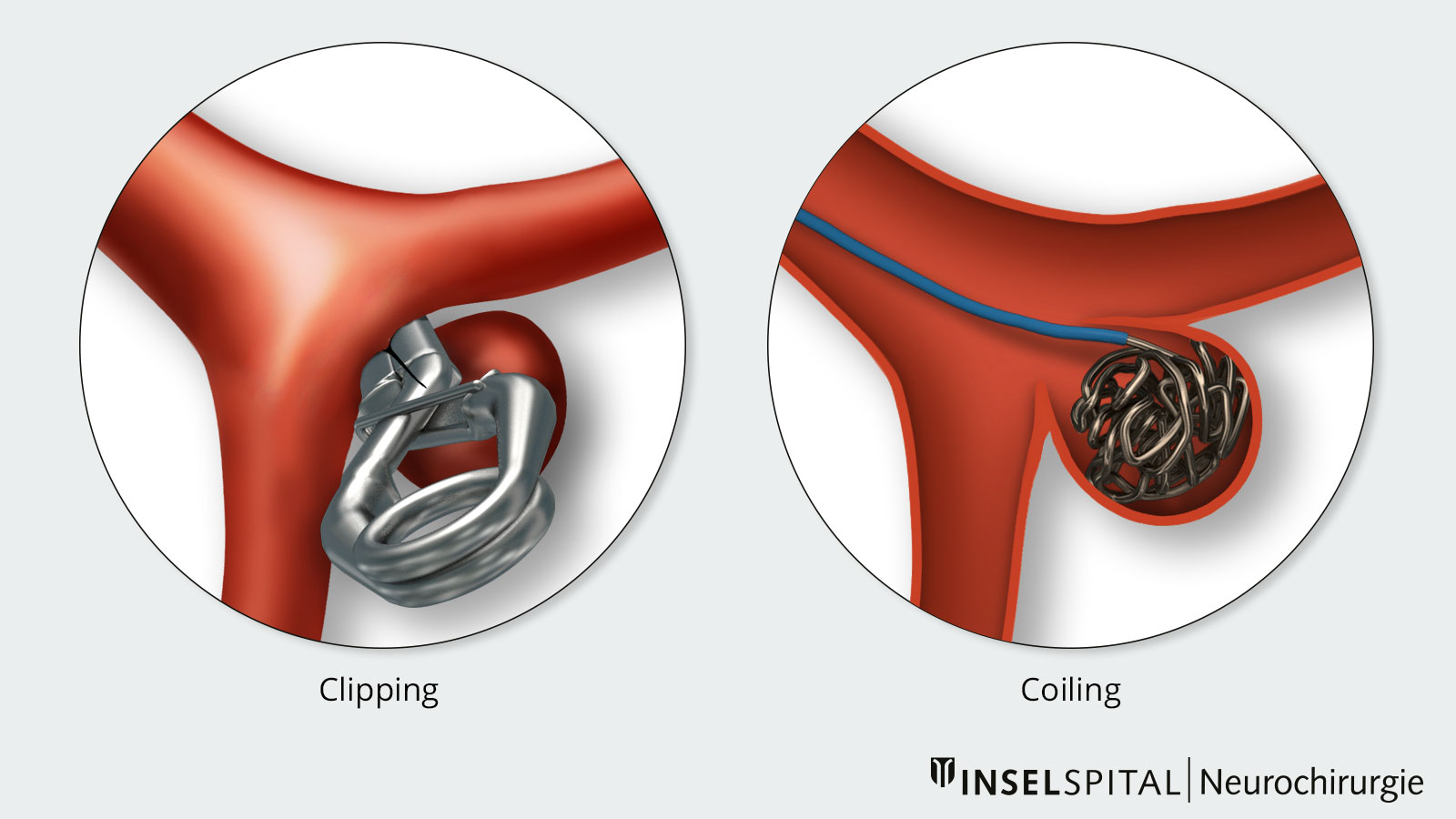
There are two different treatment techniques available for this purpose:
Clipping (microsurgical procedure)
- The aneurysm is closed off by means of a minor surgical procedure.
- This treatment is performed in neurosurgery.
Coiling (endovascular procedure)
- The aneurysm is closed off via the blood vessels using a catheter.
- This treatment is performed in interventional neuroradiology.
Microsurgical clipping
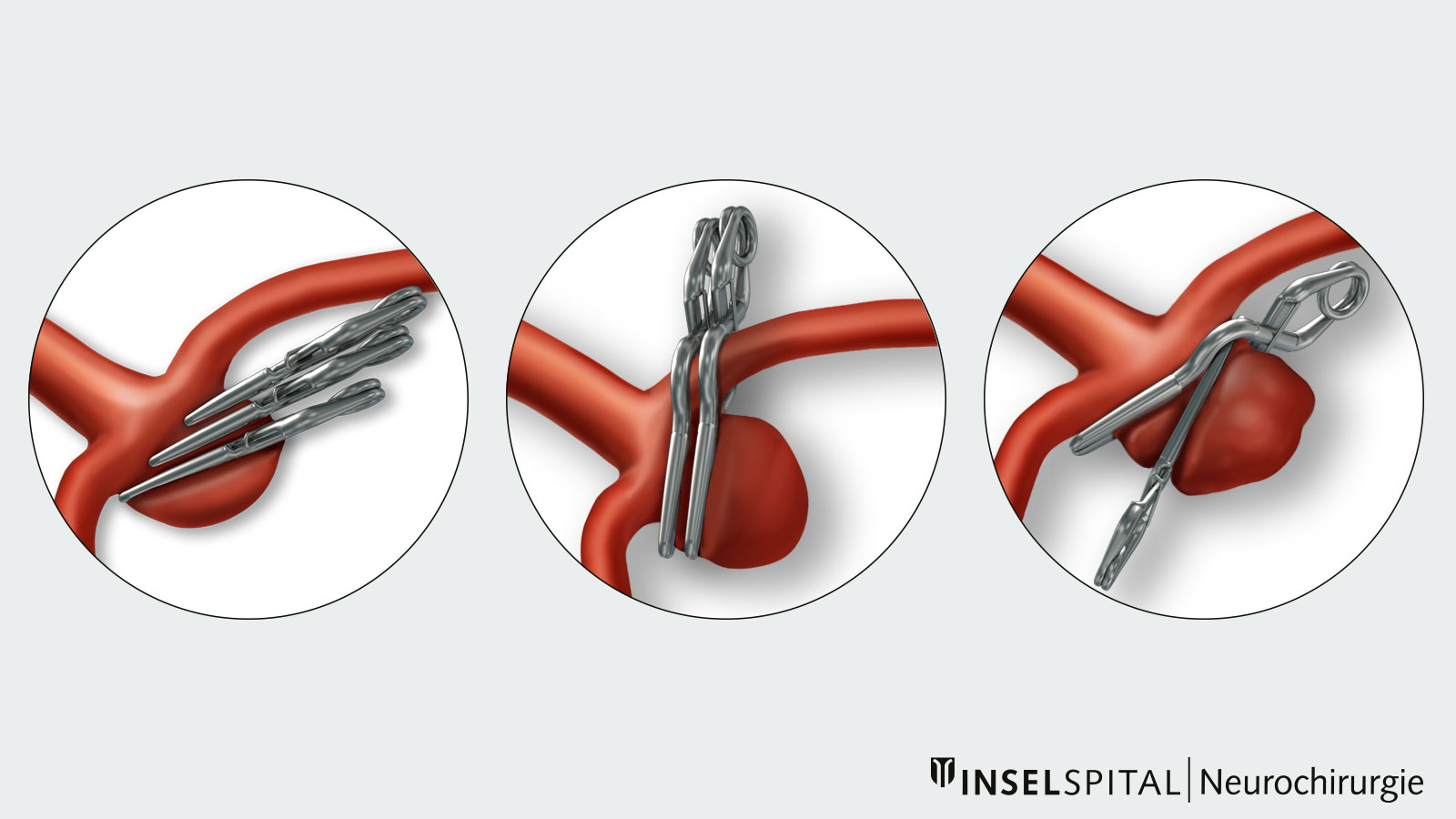
During clipping, the aneurysm is closed under an operating microscope. A small clip made of titanium or a special metal alloy is placed on the neck or base of the aneurysm and closes the aneurysm sac. Itacts like a staple, separating the aneurysm from the healthy blood vessel. A spring mechanism ensures that the aneurysm is permanently excluded from the blood flow, significantly reducing the risk of rupture. The choice of clip depends on the size, shape, and location of the aneurysm.
This method was first used in 1937 by Walter Dandy in the USA. Since then, numerous clips of different shapes and sizes have been developed to make the treatment as safe and effective as possible.
Approach
During clipping, the aneurysm is accessed via a small incision in the skin and a targeted opening in the skull in order to achieve an optimal cosmetic result. The access route is chosen so as to minimize stress on the brain.
- The surgical microscope provides high magnification and excellent lighting, allowing the vessels near the aneurysm to be identified and preserved.
- Modern techniques such as intraoperative infrared angiography *, electrophysiological monitoring, or vascular navigation increase safety and reduce stress for the patient.
- In the case of large or complex aneurysms, additional procedures are sometimes used, e.g., bypass (blood diversion), trapping (exclusion), or wrapping (redirection).
Advantages of clipping
- High closure rate of the aneurysm
- In most cases, no further follow-up examinations are necessary
Risks of clipping
The most common serious complications of clipping are:
- severe secondary bleeding
- permanent epilepsy
- vascular occlusion resulting in stroke
These risks add up to approximately 2%.
Prognosis after clipping
Approximately 97% of all aneurysms that have been carefully planned for surgery can be eliminated.
In rare cases, unforeseeable findings such as small important vessels, calcifications, or wall thickening may necessitate a switch to endovascular treatment. At our center, we always strive for maximum safety with minimal risk.
Follow-up examinations after clipping are rarely necessary. Only about 5% of patients require further observation due to small remnants, and only 1% require follow-up treatment.
Endovascular coiling
Coiling involves closing an aneurysm from the inside. Fine platinum spirals («coils») are inserted into the aneurysm via a thin catheter. These spirals fill the aneurysm, forming a blood clot (thrombosis), and the aneurysm is permanently separated from the bloodstream.
The minimally invasive endovascular treatment of cerebral aneurysms was developed in 1991 by Guido Guglielmi and colleagues with the introduction of the first electrolytically detachable coils. For the first time, it offered an alternative to microsurgical clipping *. Since the 1990s, the methods have developed rapidly: from balloon- or stent-assisted coiling to flow-diverting stents or flow diverters. Thanks to these advances, an increasingly wide range of aneurysms can now be treated gently and minimally invasively.

Approach
The procedure is performed under general anesthesia. The catheter is usually advanced through the groin artery (femoral artery) to the cerebral arteries.
- Contrast medium and X-ray fluoroscopy allow the vessels and instruments to be tracked precisely.
- Depending on the shape and size of the aneurysm, additional aids such as a balloon or a stent may be necessary to place the coils securely.
Advantages of coiling
- Minimally invasive: No major incisions in the skin or bone of the head are necessary, as access is gained via a blood vessel.
- Short recovery time: Patients are often able to resume normal activities more quickly than after open surgery.
- Wide range of treatments: Modern techniques (e.g., balloon or stent support, flow diverters) can also be used to treat more complex aneurysms.
Risks of coiling
Serious complications are rare. They occur in about 2% of treatments overall.
Possible risks include:
- Injury to the aneurysm by the coil or wire, resulting in bleeding
- Vascular occlusion, which can lead to a stroke
- Allergic reaction to the contrast agent used
Prognosis after coiling
After endovascular treatment, around 20% of aneurysms show a small residual or renewed blood flow over time. We therefore recommend regular follow-up checks:
- after 6 months
- after 24 months
- with magnetic resonance imaging or angiography if necessary
Repeat treatment after endovascular therapy is necessary in around 10% of aneurysms. If unexpected difficulties arise during the procedure, it may be necessary to switch to surgery. Here too, our center always applies the principle of choosing the treatment with the lowest risk.
Which is better: neurosurgical clipping or endovascular coiling?
Today, there is no longer any competition between surgery (clipping) and catheter treatment (coiling). Both procedures have their strengths and are used where they promise the best results.
- Clipping is technically easier for many unruptured (non-burst) aneurysms and offers a very permanent solution.
- Coiling is often less invasive for the same level of difficulty, as access is via the vessels and no surgery on the head is necessary.
In the case of an aneurysm, aneurysm-specific factors and patient-specific factors must be weighed against each other in order to determine the optimal treatment strategy for the individual patient. Rare special forms such as giant aneurysms carry a significantly higher risk and require particularly careful consideration. The decision is always made jointly by an interdisciplinary team of neurosurgeons and neuroradiologists.
In large specialized centers such as Inselspital, the risk of serious complications in typical, unruptured aneurysms of the anterior cerebral circulation that are no larger than 10 mm is approximately 2%—regardless of whether clipping or coiling is performed *, *, *.
Which factors are decisive for the choice of treatment?
In the case of an intracranial aneurysm, various factors must be taken into account and weighed against each other in order to determine the optimal treatment strategy for the individual patient.
Aneurysm size and shape
The risk of complications depends heavily on the size of the aneurysm.
- Clipping: The larger the aneurysm, the higher the risk of complications.
- Coiling: Success and occlusion rates are lower for very large aneurysms.
- Very small aneurysms (< 2 mm): Coiling is technically difficult in these cases because the aneurysm sac cannot be reliably filled with coils. In such cases, additional aids such as stents or balloons often have to be used.
- Medium-sized aneurysms: Many can be successfully closed endovascularly.
Anatomical location of the aneurysm
The location of the aneurysm in the brain plays an important role in choosing between clipping and coiling. It influences the safety and success rate of both procedures:
- Posterior cerebral vessels (e.g., basilar artery):
- Surgery is technically more difficult and associated with a higher risk in these cases. *, *
- Endovascular therapy is usually gentler and more suitable in these cases.
- Middle cerebral artery (arteria cerebri media, especially at the bifurcation):
- These aneurysms often have a wide base and vessels branching off directly from the neck of the aneurysm. They are therefore more frequently clipped.
- Endovascular treatment is possible, but often requires additional aids such as stents or balloons, which increases the risk of reopening (recanalisation) or complications.
Special aneurysms
Most aneurysms can be treated safely with clipping or coiling. However, there are rare special types that pose a challenge for both methods:
- Giant aneurysms (> 2.5 cm)
- Partially calcified aneurysms
- Partially thrombosed aneurysms (already partially filled with blood clots)
Coiling: Complete, stable occlusion is often difficult to achieve in partially thrombosed aneurysms.
Clipping: If the neck of the aneurysm is easily accessible, a clip can be a good solution. However, calcifications in the neck area can make clipping difficult or impossible.
So-called flow diverters are special stents that have expanded the possibilities of endovascular treatment in recent years. They are mainly used in large vessels supplying the brain. Their fine mesh structure redirects the blood flow in such a way that the aneurysm is gradually closed. This means that even aneurysms that were previously considered difficult or impossible to treat can now be treated.
Patient age
Several studies have shown that older patients are more likely to benefit from less invasive endovascular therapy, as it is less physically demanding. A subgroup analysis of the ISAT study shows an increased rate of disability or death in older patients with ruptured intracranial aneurysms (43.9% vs. 39.9%) when directly comparing the microsurgical treatment group with the endovascular treatment group *.
In younger patients, especially those with incidentally diagnosed, unruptured aneurysms, surgery (clipping) may have advantages because it offers a better long-term occlusion rate and complications are less common overall.
Pre-existing conditions
Patients with serious pre-existing conditions are usually treated endovascularly to reduce the risk of surgery.
Neurological condition
Endovascular therapy is also often the better choice after severe subarachnoid hemorrhage with poor clinical neurological status or vascular spasms (vasospasms).
Emergency situation
If an aneurysm bleed causes a large hematoma (blood clot), it is removed surgically as an emergency procedure. The aneurysm can be clipped directly during the same operation.
Medical expertise
When treating aneurysms, it is not only the choice between surgery (clipping) and catheter treatment (coiling) that is crucial, but also the experience of the treatment team.
Studies clearly show:
- In highly specialized centers with a large number of cases («high-volume centers»), complication rates are significantly lower *
- Experienced neurosurgeons and interventional neuroradiologists achieve better results – both for ruptured and unruptured aneurysms.
It is therefore recommended that aneurysms be treated at a supra-regional neurovascular center wherever possible, where the necessary expertise and experience are concentrated.
Our Aneurysm Center at Inselspital
- Individual assessment: Each patient is carefully examined. Neurosurgeons and interventional neuroradiologists decide together which procedure offers the greatest chance of success with the lowest risk.
- Focus on our patients: The options and recommendations are explained in a way that is easy to understand – we always determine the procedure together with the patient.
- World-class medicine: The Aneurysm Center at Inselspital offers the full spectrum of aneurysm treatment, state-of-the-art technology, and the necessary experience. With over 1,200 treatment decisions per year, Inselspital is one of the leading centers worldwide. Our experts regularly attend international conferences and help shape European recommendations.
- Research & innovation: One of our clinic's main areas of focus is the further development of aneurysm surgery. We use modern safety strategies such as
This enables us to offer aneurysm treatment of the highest standard – individualized, safe, and scientifically sound.
-
Vernooij MW, Ikram MA, Tanghe HL et al. Incidental findings on brain MRI in the general population. N Engl J Med. 2007;357:1821-1828.
-
Etminan N, Brown RD, Beseoglu K et al. The unruptured intracranial aneurysm treatment score: a multidisciplinary consensus. Neurology. 2015;85:881-889.
-
Raabe A, Nakaji P, Beck J et al. Prospective evaluation of surgical microscope-integrated intraoperative near-infrared indocyanine green videoangiography during aneurysm surgery. J Neurosurg. 2005;103:982-989.
-
Guglielmi G, Viñuela F, Sepetka I, Macellari V. Electrothrombosis of saccular aneurysms via endovascular approach. J Neurosurg. 1991;75:1-7.
-
Molyneux AJ, Birks J, Clarke A, Sneade M, Kerr RS. The durability of endovascular coiling versus neurosurgical clipping of ruptured cerebral aneurysms: 18 year follow-up of the UK cohort of the International Subarachnoid Aneurysm Trial (ISAT). Lancet. 2015;385:691-697.
-
Goldberg J, Schoeni D, Mordasini P et al. Survival and Outcome After Poor-Grade Aneurysmal Subarachnoid Hemorrhage in Elderly Patients. Stroke. 2018;49:2883-2889.
-
Molyneux AJ, Kerr RS, Yu LM et al. International subarachnoid aneurysm trial (ISAT) of neurosurgical clipping versus endovascular coiling in 2143 patients with ruptured intracranial aneurysms: a randomised comparison of effects on survival, dependency, seizures, rebleeding, subgroups, and aneurysm occlusion. Lancet. 2005;366:809-817.
-
McDonald JS, McDonald RJ, Fan J, Kallmes DF, Lanzino G, Cloft HJ. Comparative effectiveness of unruptured cerebral aneurysm therapies: propensity score analysis of clipping versus coiling. Stroke. 2013;44:988-994.
-
Seifert V, Gerlach R, Raabe A et al. The interdisciplinary treatment of unruptured intracranial aneurysms. Dtsch Arztebl Int. 2008;105:449-456.
-
Spetzler RF, McDougall CG, Zabramski JM et al. Ten-year analysis of saccular aneurysms in the Barrow Ruptured Aneurysm Trial. J Neurosurg. 20191-6.
-
Berman MF, Solomon RA, Mayer SA, Johnston SC, Yung PP. Impact of hospital-related factors on outcome after treatment of cerebral aneurysms. Stroke. 2003;34:2200-2207.